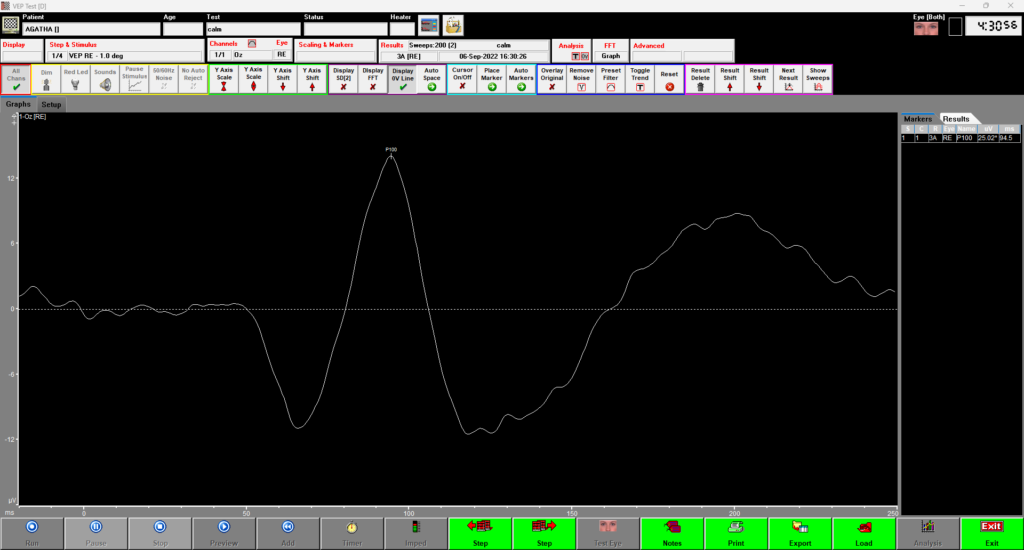
Visually evoked potentials (VEP) are visual electrophysiology tests that assess the integrity of visual pathways and evaluate optic nerve conditions involving atrophy, neuritis, compression, inflammation, demyelination, or trauma.
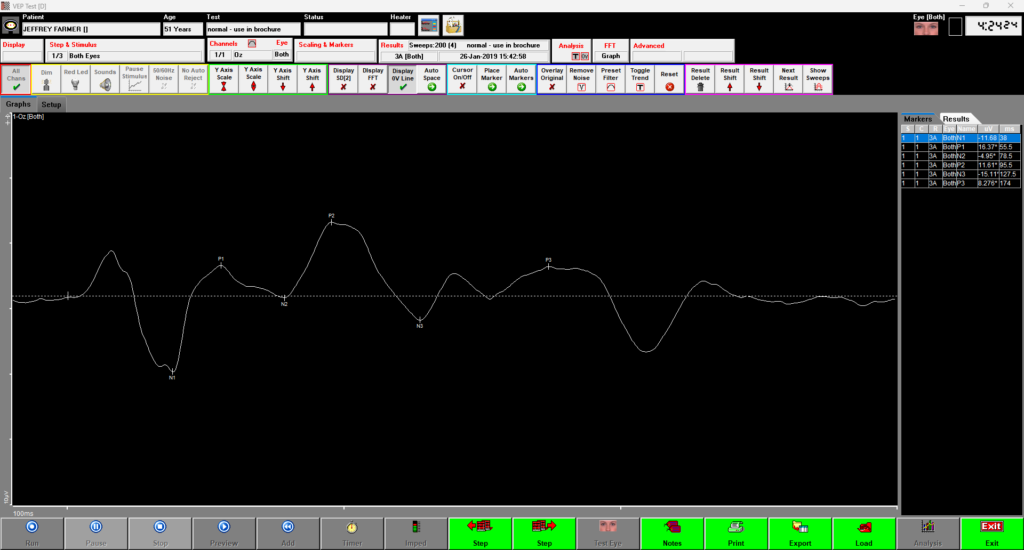
Types of VEP
Pattern VEP
Pattern VEP are typically used for individuals with good central vision and the ability to maintain fixation. For those who cannot reliably fixate, a variation known as pattern onset VEP is used.
To record a pattern VEP, electrodes are placed at the occipital cortex and forehead. The patient sits in front of a monitor with one eye patched, fixating on a central point of the screen as the pattern alternates.
Flash VEP
Flash VEPs are ideal for individuals unable to maintain fixation, such as anyone with very low vision, media opacities, under anesthesia, and young pediatric patients.
To record a flash VEP, electrodes are placed at the occipital cortex and forehead. The patient’s visual field is illuminated by bright flashes using the ColorDome® or ColorFlash.
VEP Stimuli

Standard pattern VEP
Pattern reversal VEP is the most common test due to its low inter-subject variability in both timing, amplitude, and waveform morphology.
A monitor displays a black and white checkerboard pattern of both 1.0 and 0.25 degree check sizes that reverse at a frequency of 1.5 reversals per second.
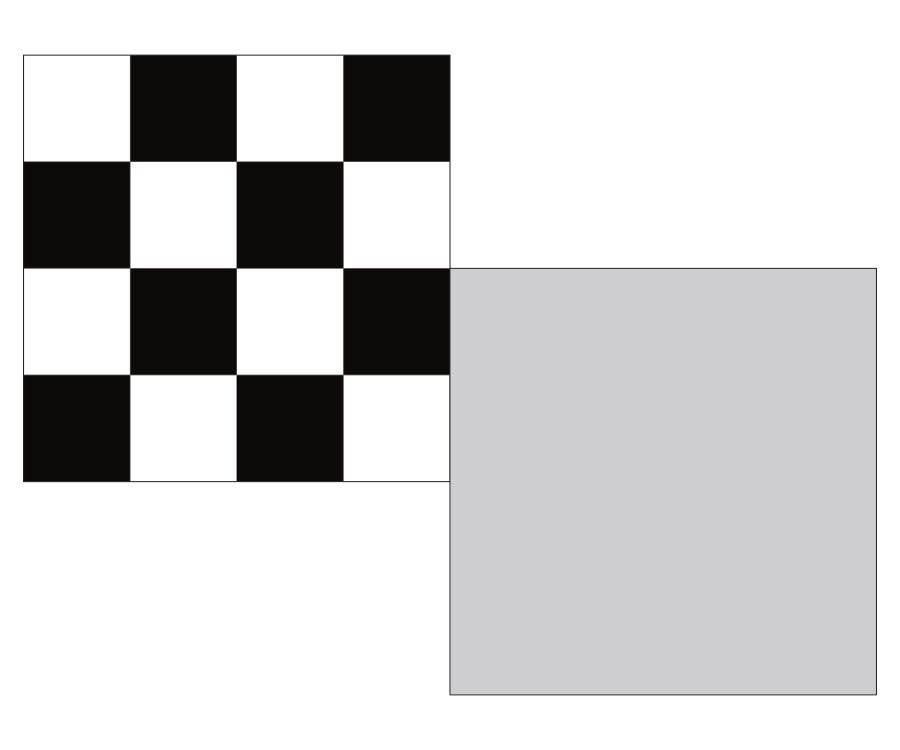
VEP for moving eyes
Pattern onset VEP are suitable for individuals with nystagmus or those suspected of malingering.
The stimulus alternates between a black and white checkerboard and a gray screen at a frequency of about 1.5 reversals per second.

VEP for obscured vision
Flash VEP are ideal for individuals unable to maintain fixation, such as anyone under anesthesia, having very low vision, or having media opacities and young pediatric patients.
Both ColorDome and ColorFlash can produce this stimulus that consists of a series of 100 flashes of white light at 3.0 cd.s/m².
VEP waveforms
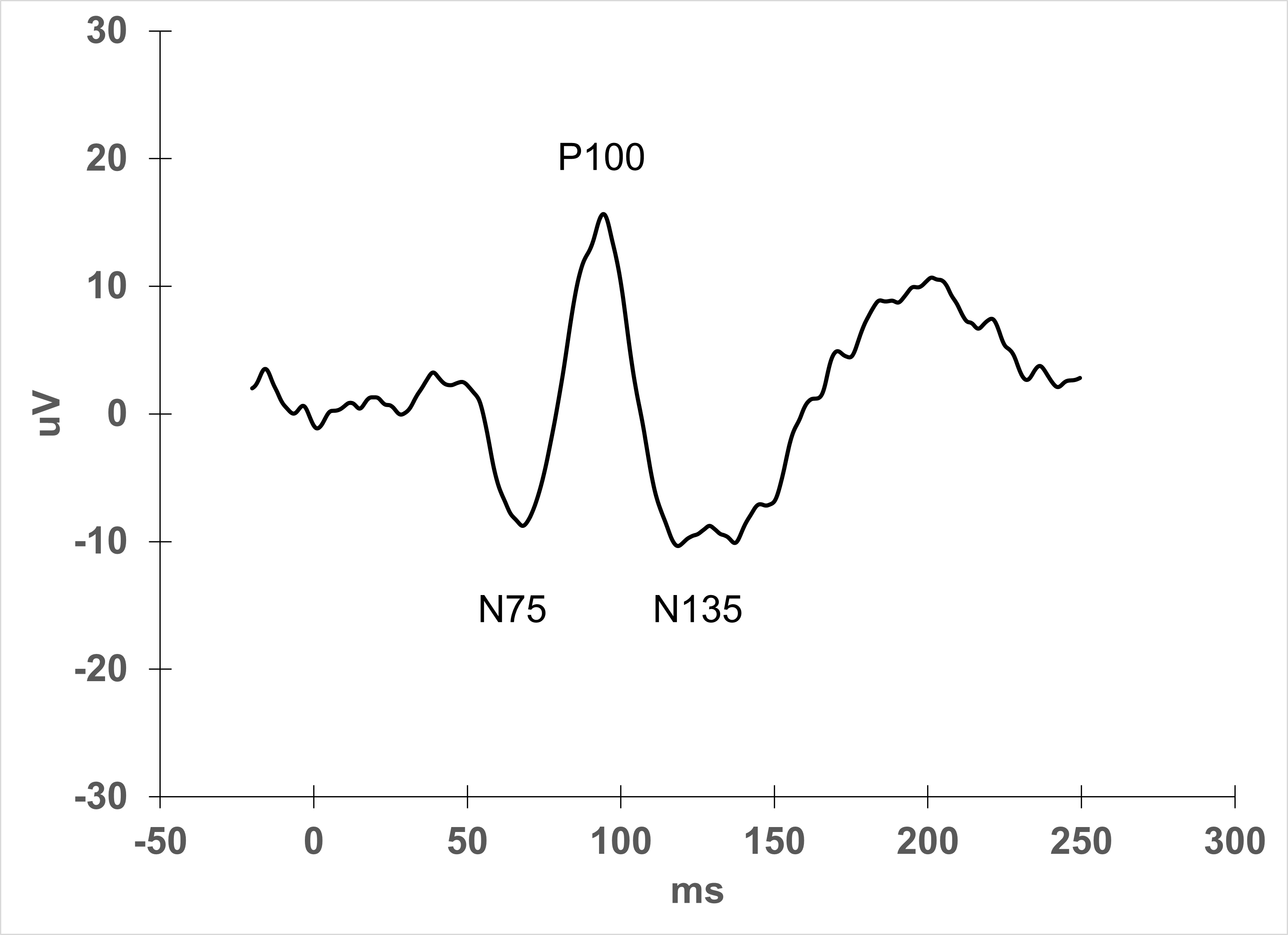
Pattern VEP
Notice the well-defined P100 peak at about 100 ms, indicating a typical normal response.
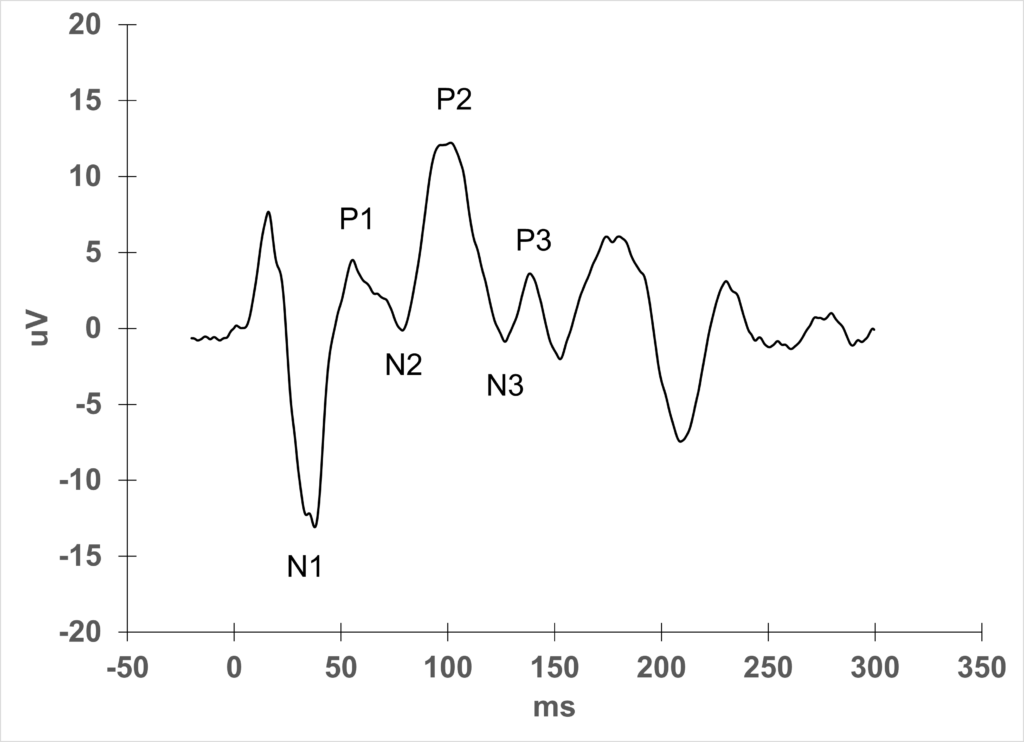
Flash VEP
You can see a normal series of peaks and valleys with a characteristic P2 marker.
Sweep VEP objective visual acuity
An objective method to estimate a person’s visual acuity
Chiasmal coefficient
A spreadsheet tool that can calculate optic nerve misrouting
References
Odom, J.V., Bach, M., Brigell, M. et al. ISCEV standard for clinical visual evoked potentials: (2016 update). Doc Ophthalmol 133, 1–9 (2016). https://doi.org/10.1007/s10633-016-9553-y
